
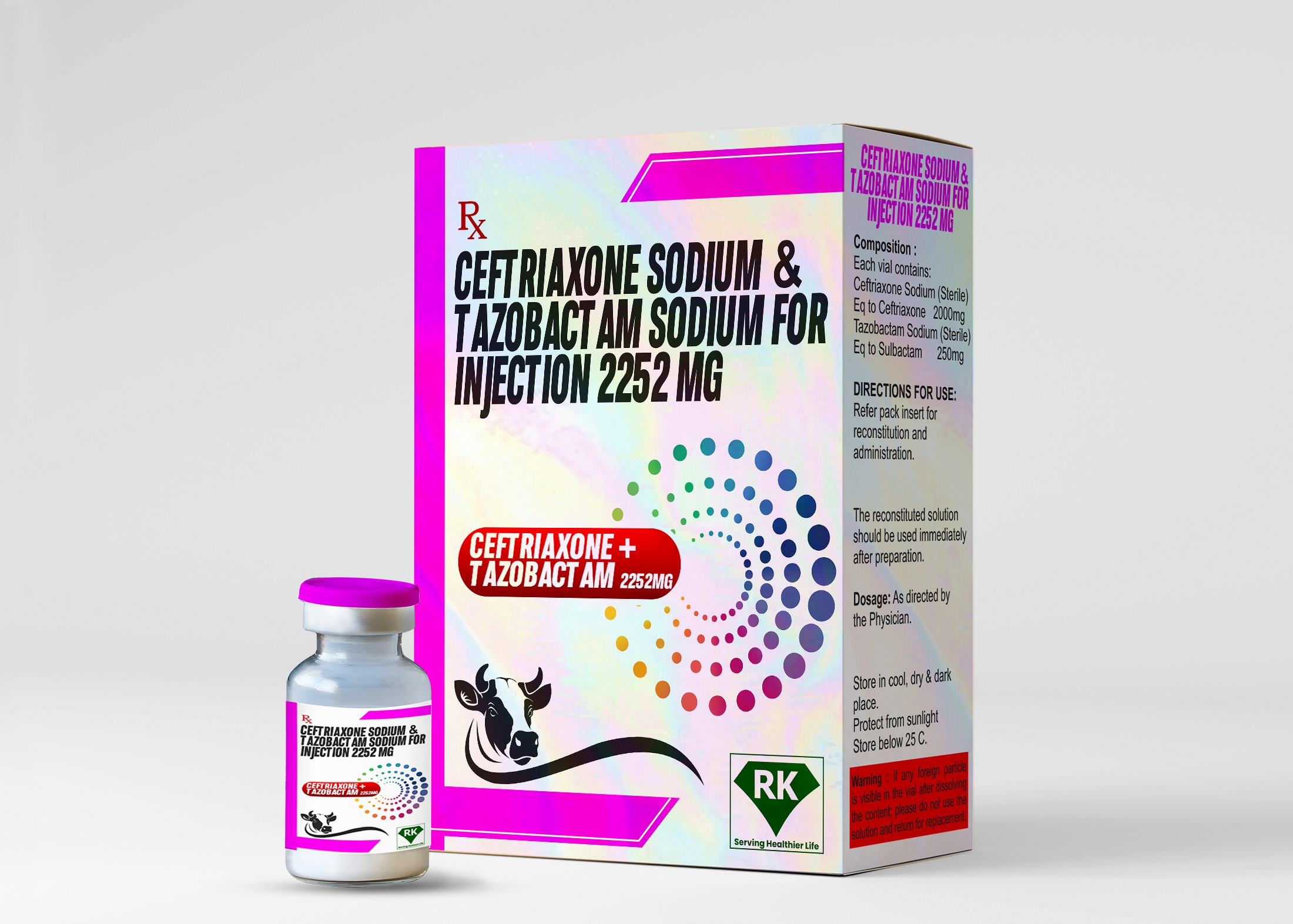
Ceftriaxone Sodium & Tazobactam Sodium for Injection 2252mg
R K Lifecare INC: Trusted Manufacturer and Wholesaler of Ceftriaxone Sodium and Tazobactam Sodium for Injection 2252mg
At R K Lifecare INC, we are proud to be one of the leading Ceftriaxone Sodium and Tazobactam Sodium for Injection 2252mg manufacturers in India, providing essential medicines that are critical in the treatment of a variety of serious bacterial infections.
As a reliable and trustworthy Ceftriaxone Sodium Injection manufacturer in India and Tazobactam Sodium Injection manufacturer in India, we are dedicated to ensuring that healthcare providers have access to high-quality injectable antibiotics that can effectively deal with infections that threaten patients' lives.
With the rise of antibiotic-resistant pathogens and the growing need for potent combination therapies, the Ceftriaxone Sodium and Tazobactam Sodium for Injection 2252mg combination offers healthcare providers a reliable and effective solution for managing infections.
The formulation of Ceftriaxone Sodium and Tazobactam Sodium works synergistically to treat a wide range of bacterial pathogens, including those resistant to many other antibiotics, making it an indispensable tool in the fight against serious bacterial infections.
As one of the most reputed Ceftriaxone Sodium Injection wholesalers in India, We understand the importance of providing hospitals, clinics, and healthcare institutions with a consistent and reliable supply of Ceftriaxone Sodium and Tazobactam Sodium for Injection 2252mg. We are proud to serve as Ceftriaxone Sodium Injection distributor in India and Tazobactam Sodium Injection distributor in India, and we work diligently to ensure that our products are available across the country, including areas such as Ceftriaxone Sodium Injection in Delhi NCR.
At R K Lifecare INC, We also collaborate with a network of Ceftriaxone Sodium Injection dealers and Tazobactam Sodium Injection dealers, allowing us to expand our reach and make these life-saving injections available to even more healthcare professionals in need of quality antibiotics.
Our commitment to quality and safety drives every aspect of our manufacturing and distribution processes. As one of the leading Ceftriaxone Sodium Injection suppliers in India and Tazobactam Sodium Injection suppliers in India, we focus on manufacturing injections that meet or exceed the highest industry standards. Our team ensures that every batch of Ceftriaxone Sodium and Tazobactam Sodium Injection 2252mg is rigorously tested for potency, purity, and safety before it reaches healthcare providers.
This dedication to quality is what makes us a trusted supplier for hospitals, clinics, and pharmacies across India and other international markets. We understand that healthcare providers rely on our products to save lives, which is why we maintain the highest standards at every step of our production and distribution processes.
The combination of Ceftriaxone Sodium and Tazobactam Sodium Injection 2252mg is used to treat a wide range of bacterial infections, including those affecting the respiratory tract, urinary tract, skin and soft tissues, and the inner parts of the abdomen. By combining the third-generation cephalosporin antibiotic ceftriaxone with tazobactam, a beta-lactamase inhibitor, the drug effectively combats bacterial strains that are otherwise resistant to conventional antibiotic therapy.
This makes Ceftriaxone Sodium and Tazobactam Sodium Injection 2252mg an essential therapeutic option for patients suffering from severe and life-threatening bacterial infections. This is particularly important in intensive care settings, where timely and effective antibiotic treatment is crucial for patient survival. Manufacturing Process and Quality Assurance
At R K Lifecare INC. We follow a meticulous process to ensure that each vial of Ceftriaxone Sodium and Tazobactam Sodium 2252mg for Injection meets the highest standards of quality and safety. The production process begins with the careful procurement of raw materials.
We source our active pharmaceutical ingredients (APIs) only from certified suppliers who adhere to global quality standards. This ensures that the basic ingredients used in the manufacturing process are of the highest purity and potency. Each batch of Ceftriaxone Sodium and Tazobactam Sodium is thoroughly tested before it enters the production line, to ensure that it meets the strictest specifications for quality and consistency.
Once the raw materials are sourced and tested, they are carefully blended to form the Ceftriaxone Sodium and Tazobactam Sodium Injection 2252mg solution. The manufacturing and blending process is carried out under rigorous conditions to ensure uniformity and effectiveness in the final product. Each component of the solution is measured in the correct amount and mixed in a controlled environment to create the correct amount of dosage. This step is crucial in ensuring that the final product provides consistent therapeutic results and is free of any contaminants.
After the manufacturing and blending process, the solution undergoes sterile filtration. This step is essential in removing any potential microbial contaminants that could compromise the sterility of the injection. The solution is then filled into vials in aseptic conditions, ensuring that the final product remains free from contamination throughout the entire manufacturing process.
We use state-of-the-art filling equipment to ensure that each vial contains the exact amount of Ceftriaxone Sodium and Tazobactam Sodium 2252mg for Injection, allowing healthcare providers to deliver accurate and reliable dosages.
For some formulations, lyophilization (freeze-drying) is used to improve product stability and shelf life. Lyophilization involves freezing the solution and then removing the water content under vacuum, which helps preserve the active ingredients while maintaining their effectiveness over time. This is particularly useful for injectable medications that may be sensitive to temperature fluctuations and environmental conditions. Once the lyophilization process is complete, the vials are closed and sealed to ensure they remain sterile and safe during transportation and storage.
Quality Control (QC) and Quality Assurance (QA)
Quality is paramount in the production of pharmaceutical products, and at R K Lifecare INC, We take great care to ensure that our Ceftriaxone Sodium and Tazobactam Sodium for Injection 2252mg meets the highest industry standards. Our Quality Control (QC) team rigorously tests every batch of the drug to ensure that it meets strict specifications for potency, purity, sterility, and safety. This includes testing for microbial contamination, ensuring that Ceftriaxone Sodium and Tazobactam Sodium for Injection 2252mg is free from harmful pathogens that could endanger patient health.
In addition to regular QC tests, our Quality Assurance (QA) team ensures that every step of the manufacturing process complies with regulatory requirements and international standards. We keep detailed records of each production batch, including the source of raw materials, manufacturing conditions, and the results of all quality tests. This comprehensive approach ensures that we consistently make safe and effective medicines.
Uses and Side Effects
Ceftriaxone Sodium and Tazobactam Sodium for Injection 2252mg is a combination antibiotic used to treat a wide variety of bacterial infections, including:
Respiratory tract infections (e.g., pneumonia, bronchitis)
Urinary tract infections
Intra-abdominal infections (e.g., peritonitis)
Skin and soft tissue infections
Bacterial septicemia
Gynecological infections
The combination of ceftriaxone sodium, which targets the bacterial cell wall, and tazobactam sodium, which inhibits the beta-lactamase enzyme, makes this medicine effective against a wide range of pathogens, including some resistant strains.
However, like all medicines, Ceftriaxone Sodium and Tazobactam Sodium for Injection 2252mg can cause side effects. Common side effects include:
Reactions at the injection site (pain, redness, swelling)
Nausea and vomiting
Diarrhea
Elevated liver enzymes
More serious side effects may include allergic reactions, such as rash, itching or swelling, and in rare cases, anaphylaxis. It is important that healthcare providers monitor patients closely during treatment and be prepared to manage any adverse reactions immediately.
Labeling and Packaging
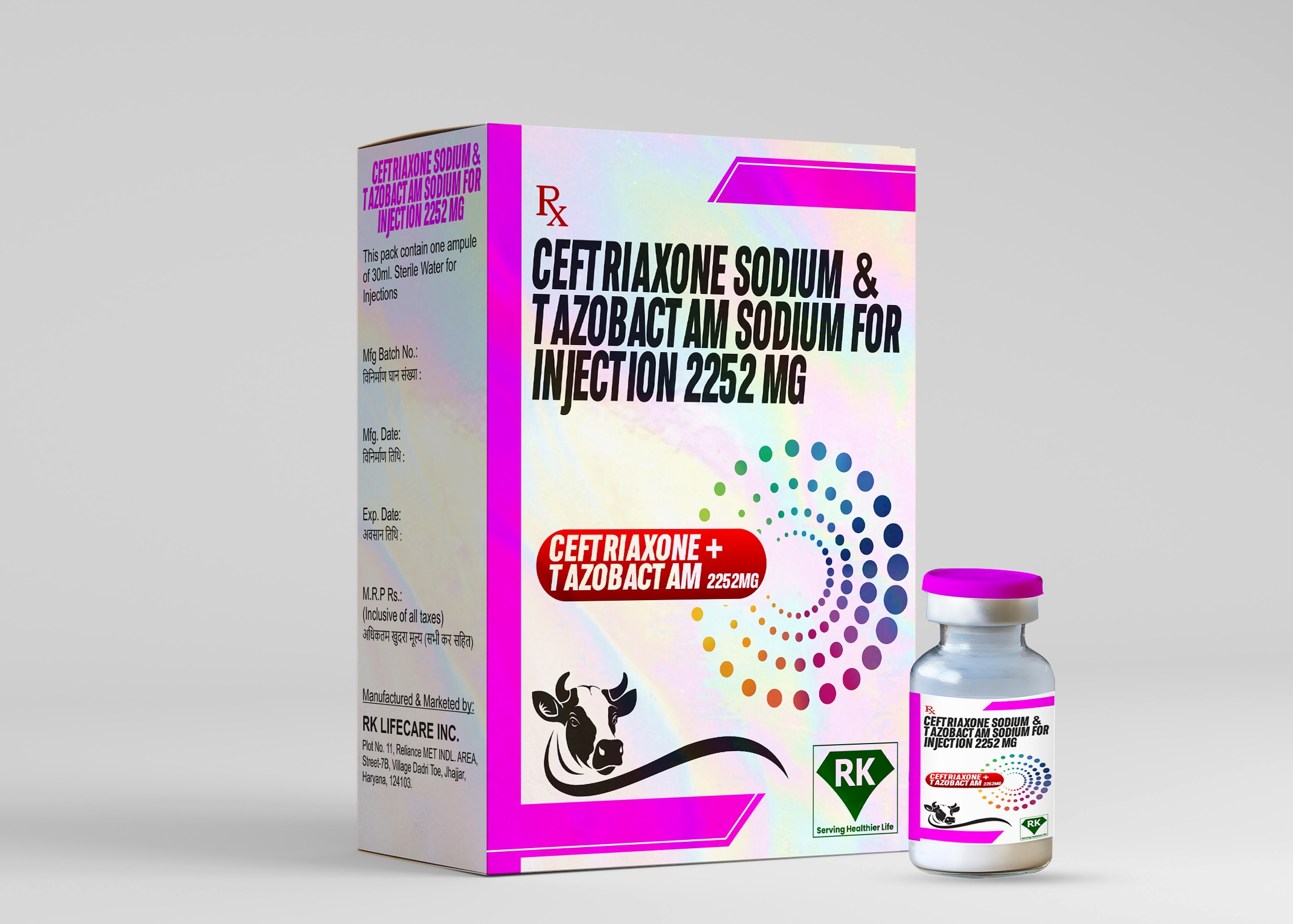
The labeling and packaging of Ceftriaxone Sodium and Tazobactam Sodium Injection 2252mg is done in compliance with regulatory standards to ensure that healthcare providers have all the necessary information to use the medicine safely and effectively. Important details such as dosage strength, instructions for use, and potential side effects are labeled on each vial. The packaging is designed to protect the product from contamination and damage during transport, ensuring that the medicine reaches healthcare providers in optimal condition.
Storage and Warehousing
Proper storage and warehousing of Ceftriaxone Sodium and Tazobactam Sodium Injection 2252mg is essential to maintain the stability and effectiveness of the product. At R K Lifecare INC, We ensure that our storage facilities are temperature-controlled, following the recommended storage conditions for injectable medications. This prevents degradation of active ingredients and ensures that the product remains effective until its expiration date. Our warehouses are equipped with inventory management systems to track the movement of each batch of the product, ensuring that it is delivered in a timely and efficient manner.
Distribution and Supply Chain
As a Ceftriaxone Sodium Injection supplier in India and Tazobactam Sodium Injection supplier in India, we have developed an efficient distribution and supply chain to ensure that Ceftriaxone Sodium and Tazobactam Sodium Injection 2252mg is available to healthcare providers across India and beyond. Our supply chain is optimized for speed and efficiency, ensuring that orders are processed and delivered promptly. We work closely with a network of Ceftriaxone Sodium Injection distributors in India and Tazobactam Sodium Injection distributors in India to ensure that our products reach hospitals, clinics and pharmacies across the country. Through this extensive network, we are able to ensure that life-saving medicines reach those who need them the most.













.jpg)